Innovating means not only inventing something new, but also improving the existing.
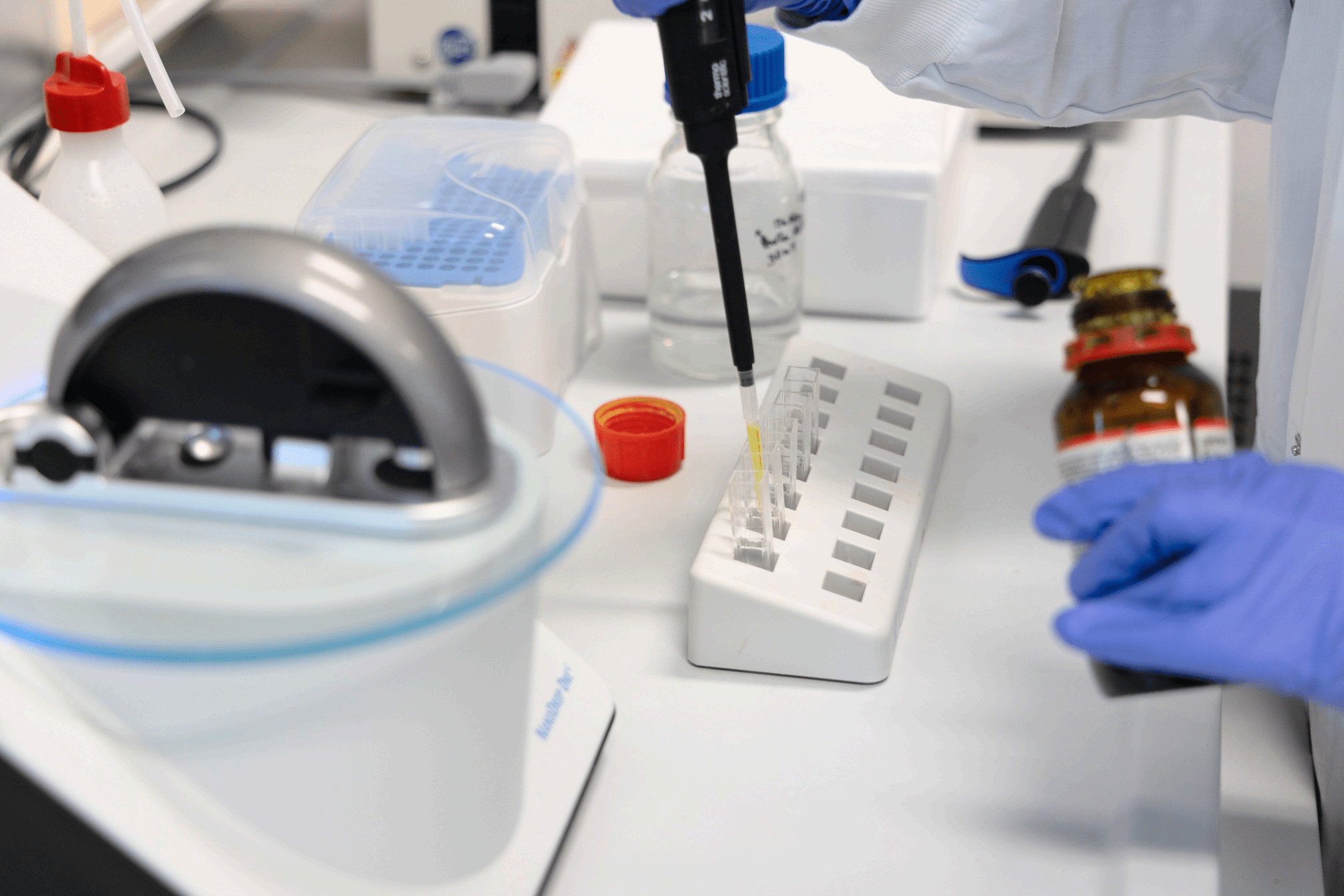
Our Research and Development (R&D) department is organised into two specific areas: pharmaceutical technology and biotechnology.
Our research activities focus on studying new pharmaceutical technologies and formulations to improve the bioavailability, tolerability and use of active ingredients, thereby promoting patient adherence to therapy. through our development activities, we aim to create and refine existing formulations and technologies in order to ensure greater efficacy and ease of use in the pharmaceutical and nutraceutical fields and in the area of medical devices.
We explore solutions that intercept physician-patient needs in a timely manner.
Our R&D laboratories in Pavia, renovated in 2019, are engaged in various projects involving an almost entirely female team of experts from various fields, who work in synergy, combining pharmaceutical research with nutraceutical solutions to address health and wellness challenges. We are committed to innovating therapeutic and nutritional solutions with a focus on medicines, supplements and medical devices.




Retard technology

One example of development is the retard system that Savio has applied to one of the main drugs for the treatment of type 2 diabetes mellitus (T2DM), metformin, improving therapeutic management and the quality of life of diabetic patients, without ruling out the possibility of applying the same system to other drugs that could benefit from it. In addition, the group is constantly engaged in the study and renewal of the analytical methods used for the qualitative and quantitative analysis of the products in its portfolio, in order to keep up with the times and ensure cutting-edge analysis procedures. Savio’s R&D Department has obtained the publication of a national patent for the development of a two-dimensional analytical method for the determination of vitamin D3 in oily formulations, developed in collaboration with the University of Pavia (Patent issued by UIBM – Italian Patent and Trademark Office no. 102023000007014).
Savio Probiotics

The company focuses on characterising specific probiotic strains and their beneficial activities in collaboration with the University of Naples. Preclinical in vivo studies [1,2,3] have shown that our spore-forming strains [Bacillus subtilis SF-106 (DSM 34548) and Bacillus clausii SF-174 (DSM 34549) not only modulate the gut microbiota, increasing the amount of “good” bacteria in our intestines, but also have anti-inflammatory and antioxidant properties. Oxidative stress is the trigger for increasingly common diseases such as metabolic syndrome, pre-diabetes and MASLD (metabolic dysfunction-associated steatohepatitis, formerly known as NAFLD – non-alcoholic fatty liver disease), for which there is still no defined pharmacological approach and for which the strong involvement of the microbiota is well known. The aim of our research is to maintain the balance of the gut microbiota in order to prevent metabolic diseases.
In this regard, two supplements based on spores from the Savio probiotic strain have recently been launched on the market: Biliver for patients with MASLD and Promet12 for patients with T2DM who have vitamin B12 malabsorption.
Among the most recent technologies developed for the formulation of supplements, which were the subject of a recently published scientific study[4], the technology called Spore Surface Display (SSD) is particularly noteworthy. SSD is a non-recombinant system that uses probiotic spores, forms of bacterial cell resistance, as delivery vehicles for molecules of biopharmaceutical interest. The technology allows the beneficial properties of spores to be combined synergistically with those of bioactive molecules.
The Savio R&D Department is also active in improving the effectiveness of hyaluronic acid viscosupplementation (in the form of medical devices) in the treatment of osteoarticular diseases.
The Savio Group is aware of the economic burden that chronic disease management places on the National Health Service. For this reason, part of our R&D is dedicated to finding solutions that combine the essential quality required of medicines with the optimisation of treatment costs. Examples of this are some of the group’s brands used for the control of dyslipidaemia and the treatment of osteoporosis, which offer packs with a greater number of tablets and therefore last longer and have a lower cost per treatment, making them convenient for the doctor, the patient and healthcare expenditure in terms of both economics and adherence to therapy.
References:
[1] Saggese A, Barrella V, Porzio AD, Troise AD, Scaloni A, Cigliano L, Scala G, Baccigalupi L, Iossa S, Ricca E, Mazzoli A. Protective role of cells and spores of Shouchella clausii SF174 against fructose-induced gut dysfunctions in small and large intestine. J Nutr Biochem. 2024 Nov;133:109706. doi: 10.1016/j.jnutbio.2024.109706. Epub 2024 Jul 23. PMID: 39053859.
[2] Vittoria M, Horwell E, Bastoni D, Saggese A, Baccigalupi L, Cutting SM, Ricca E. Bacillus subtilis SF106 and Bacillus clausii SF174 spores reduce the inflammation and modulate the gut microbiota in a colitis model. Benef Microbes. 2024 Jun 14;15(4):343-355. doi: 10.1163/18762891-bja00016. PMID: 38879179.
[3] Di Porzio A, Barrella V, Saggese A, Baccigalupi L, Cigliano L, Ricca E, Iossa S, Mazzoli A. Fructose-Induced Impairment of Liver and Skeletal Muscle Metabolism Is Prevented by Administration of Shouchella clausii Spores by Preserving Mitochondrial Function and Insulin Sensitivity. Mol Nutr Food Res. 2025 Jun;69(11):e70063. doi: 10.1002/mnfr.70063. Epub 2025 Apr 10. PMID: 40207597; PMCID: PMC12128003.
[4] Corona R, Bontà V, Baccigalupi L, Ricca E. Probiotic Spores of Shouchella clausii SF174 and Displayed Bromelain Show Beneficial Additive Potential. Int J Mol Sci. 2025 Jan 23;26(3):942. doi: 10.3390/ijms26030942. PMID: 39940711; PMCID: PMC11817855